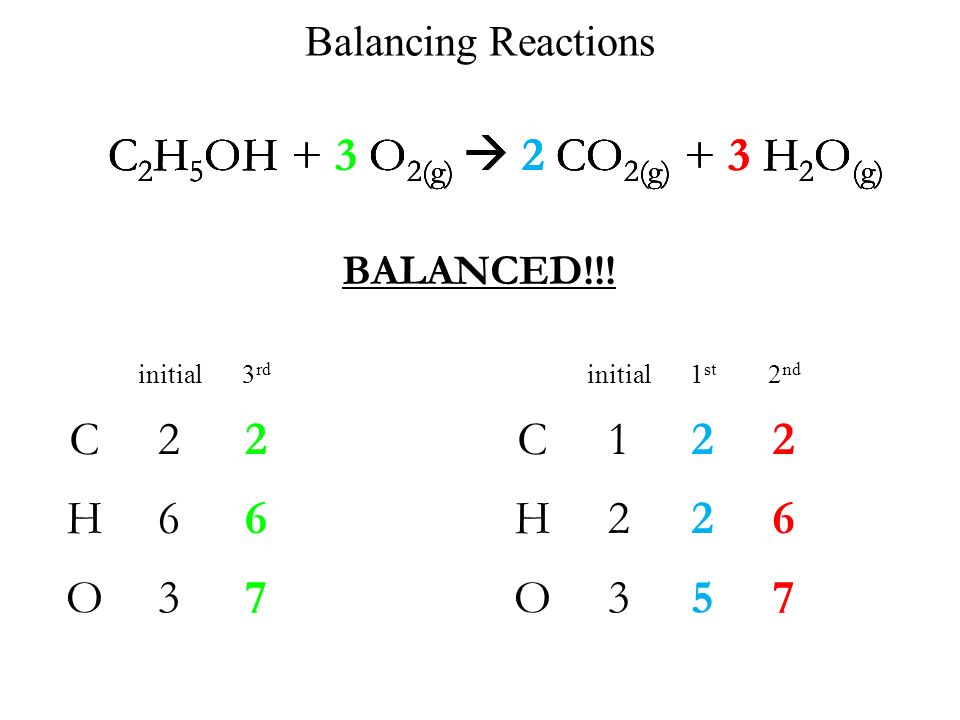 
Looking at it again, we gots both LHS and RHS neutral, LHS and RHS, #2xxS#, #3xxO#, and #2xxH#.so balanced with respect to mass and charge. #underbrace(S_2O_3^(2-) +2H^(+))_("sulfurous acid"*H_2SO_3) rarr S +H_2O +SO_2#Īgain, if mass and charge ARE NOT BALANCED, then we CANNOT accept it as a representation of chemical reality. #2S_2O_3^(2-) +4H^(+) rarr 2S +2H_2O +2SO_2#Īnd of course we could halve this, as per Nam D.'s suggestion. We add #(i) + (ii)# to remove the electrons. 
Are they balanced? Don't trust my arithmetic. zerovalent sulfur, and oxidized to #stackrel(+IV)"SO"_2#. an average oxidation state of #(-II+VI)/2=+II#. Thiosulfate, #S_2O_3^(2-)# has formal #stackrel(-II)S# and #stackrel(+VI)S# oxidation states, i.e.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
